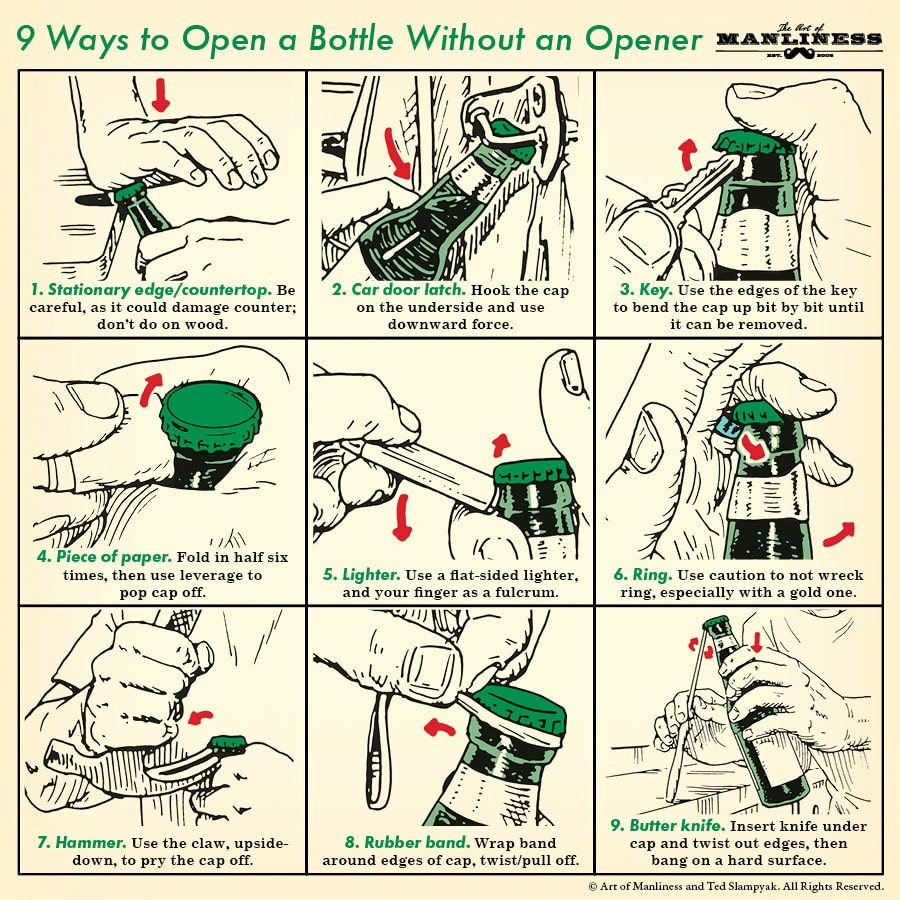
An important part of manhood has always been about having the competence to be effective in the world — having the breadth of skills, the savoir-faire, to handle any situation you find yourself in. With that in mind, each Sunday we’ll be republishing one of the illustrated guides from our archives, so you can hone your manly know-how week by week.
It is one of the planet’s great ironies that a person can be lost at sea, surrounded by water, and die of dehydration. The reason saltwater is so harmful to drink is not that you can’t drink salt. We drink salty liquid, like soups and stews, all the time, and our kidneys have no problem filtering out the salt and processing it. But that process works because we also drink lots of freshwater, which ultimately dilutes the total concentration of salt in our bodies. It’s a balance.
When you drink just saltwater, you throw that balance way off-kilter. Your kidneys go into overdrive, pulling salt out and forcing you to urinate. Every time you pee, you lose water, and no matter how fast you drink, you’ll never be able to catch up.
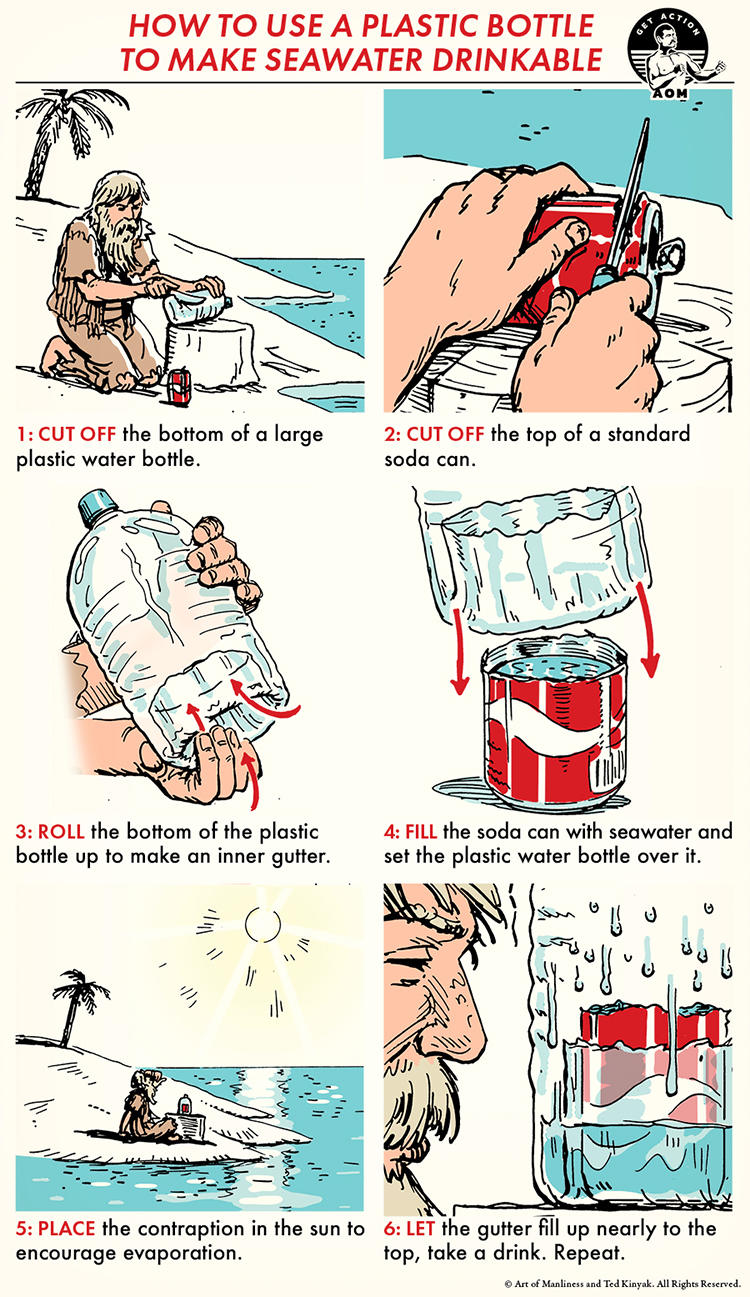
In survival scenarios, desalination (pulling the salt out of water) is the only way to make seawater safe enough to drink. The simplest form of desalination is basic evaporation. Think back to your elementary science class: You were probably taught that the sun heats surface water on the ocean, which causes evaporation. When water evaporates, it leaves the salt behind. The moist air rises, cools, creates clouds, and, if there’s enough of it, rain.
The desalination method below relies on that process of evaporation and condensation to help you capture freshwater in a container to drink. All you need is a large plastic bottle and a soda can. Keep in mind that your plastic bottle must have a lid. Otherwise, the evaporated seawater will escape through the top.
Like this illustrated guide? Then you’re going to love our book The Illustrated Art of Manliness! Pick up a copy on Amazon.